Protocols and IRB
PhaseⅠ
PhaseⅡ
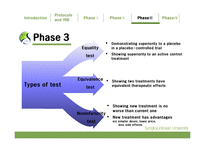
PhaseⅢ
PhaseⅣ
Clinical trial is limited to human, therefore it is the precondition that ensuring ethicality with scientific validity of study.
Necessity of Clinical test
- Developing a new drug for Korean inherited trait
- Offering the earliest opportunity to patients
- Strengthening the capability of researchers
- Clinical trial has high added value
It is a plan that recorded specific details
· How clinical trials should be performed
Including all aspects of the research study
· participant inclusion / exclusion criteria
· test schedules and procedures
· medications and their dosages
· methods used to analyze the data collected
- By protocols, experiment could get validity
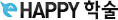



















 분야
분야


